All you need to know about Linear Alkylbenzene (LAB)
Introduction
Increasing population in different parts of the world and changes in the lifestyle of the people – especially in developing countries – have changed the traditional way of life forever. Today, in many parts of the world, such as China and India, we witness changes in people’s lifestyles. Over the past decades, the industrialization of these countries has led to an increase in middle-class households. Naturally, one of the most critical issues facing such families is health. More importantly, the requirement of developed countries to improve the technologies used to maintain their health standards makes a fascinating market for research and development of technologies related to improving the quality of health products, including detergents. This point is where the name of one of the petrochemical products comes into play: linear alkylbenzene (LAB). one of the petrochemical industry’s most essential products.
LAB is the intermediate for producing various detergents, from Household detergents such as soap, shampoo, and similar products to industrial detergents in a world that moves towards industrialization with incredible speed. Add to the changing lifestyle of the people, the issue and the need for more and more people who use different types of detergents to realize the importance of the petrochemical industry, primarily linear alkylbenzene. In this article, we examine the lAB structure, the reasons for its popularity, and its production methods. Finally, we will briefly review the LAB offered by KIA Middle East Company.
Chemical structure of linear alkylbenzene (LAB)
This section briefly explains the two words, alkyl, and benzene.
What is Alkyl?
If we separate one hydrogen atom from the molecule of the famous family of alkane hydrocarbons, the rest is called the alkyl group. The general formula for the group of alkyls is CnH2n + 1 because it has one more minor hydrogen atom than the parent alkane (CnH2n + 2). These groups are named by removing (-ane) from the corresponding alkane name and replacing it with (-yl), all known as alkyl groups.
Benzene
Benzene (C6H6) is a hydrocarbon family. An organic chemical compound with a cyclic arrangement called the benzene ring is also found in many combinations, including aspirin and the explosive trinitrotoluene. Adding it to the gasoline increases the octane number of the product and reduces the chance of engine crashes.
Benzene, or cyclohexatriene, is a cyclic molecule in which six carbon atoms form a regular hexagonal structure attached to a hydrogen atom. Covalent bonds between carbon atoms are one-to-one, simple, and dual.
In the past, benzene was obtained by heating coal and then converting its vapor into liquid. Today, however, it is extracted in large quantities of crude oil. Benzene is a colorless, fragrant liquid that burns with a yellow flame with soot, evaporates quickly, and has exceptionally high flammability.
This material is used in the industrial production of plastics, resins, nylon, synthetic rubber, lubricating oils, dyes, cleaners, and pesticides. This liquid is also used in the preparation of detergents and paints. All benzene derivatives can be considered aromatic organic substances like naphthalene or toluene. Benzene, one of the essential products of the petrochemical industry and is used to produce millions of kilograms of more complex structures such as ethylbenzene, is becoming more limited due to its toxicity.
Linear alkenebenzene (LAB)
After getting taught the alkyl and benzene groups, we will be taught the chemical composition of linear alkylbenzene (LAB) and its chemical formula.
Linear alkyl benzene consists of a benzene ring and a linear alkyl group attached to it. The alkyl group attached to the linear alkylbenzene is a linear organic hydrocarbon group. So, it consists of two parts, a linear alkyl chain and a benzene ring. This chemical product is in the group of aromatic hydrocarbons. Linear alkylbenzene, known as LAB, is represented by the chemical formula C6H5CnH2n + 1.
CnH2n + 1 is a branched-chain mainly produced as an intermediate for making surfactants for use in detergents. Typically in the chemical formula of alkylbenzene, the linear n is between 10 and 16. Heavy sections such as C12 -C13, C12-C15, and C10-C13 are prepared for dishwashing liquid, car wash shampoo, car detergents, and sanitary detergents.
How to produce LAB?
As mentioned above, Linear alkyl benzene consists of a linear chain and a benzene ring. Kerosene is the raw material of high purity linear paraffin, eventually hydrogenated to linear olefins. As a result, linear olefins are combined with benzene to produce linear alkylbenzene. Applications of this product are divided into two groups: linear alkylbenzene sulfonate (LAS) and non-surfactant.
LAS is used in detergents to produce soaps, powders, and other industrial and commercial cleaners, and non-surfactants are used in agriculture, humidifiers, and dye industries. LAB now accounts for almost the entire production of alkylbenzene sulfonates, and its demand enhanced from 1 million metric tons per year in 1980 to 1.8 in 1990. The production capacity reached 2.4 million tons per year by 2000.
Why is LAB so important?
Because LAB does not have any replacement.
Linear alkylbenzene is a highly cost-effective and biodegradable intermediate in the production of detergents and surfactants.
Two characteristics, each of which is considered a golden card for a petrochemical product.
Living in a world where persevering the environment has become the top priority of many producers and consumers, making LAB’s biodegradable feature important.
Since the 1960s, LABs have emerged as the dominant precursor of biodegradable detergents.
The method of producing LAB has almost replaced the old branched alkylbenzene technology surfactants based on enhanced biodegradation and cost-effectiveness. Today, the method of choice is the creation of normal paraffin to normal olefin, followed by alkylation of benzene to form LAB. But how?
Different methods that the petrochemical engineering uses are based on catalysts. The main techniques are the Hf process and the Detal process. But At first:
Why Using a catalyst?
The activation energy between the reaction of benzene and olefine to make LAB is very high. Therefore, high temperature and pressure are needed.
So in 1968, it was the first time HF was used to produce LAB from linear alkanes in the Pacol unit.
The LAB is obtained by benzene alkylation through the Friedel–Craft catalyst in the presence of aluminum chloride or HF. Linear alkenes are obtained by dehydrogenation of alkanes. HF performs the two alkylation processes, but most alkylation in liquid products is performed by the Friedel-Craft method in the presence of AlCl3.
N-Paraffin of required C-chain length is extracted from Kerosene in the Molecular Extraction Unit. It is dehydrogenated into corresponding olefins in PACOL and DeFining unit. Alkylation of benzene is done with these mono olefins to form Linear Alkyl Benzene. Some heavy Alkylate generated during the process is separated by fractionation.
Many solid catalysts are used for the alkylation process. Still, one of the most critical problems of solid catalysts is the chance of forming carbon deposits due to possible diolefin present in the feed on the active pores of the catalyst and as a result of catalyst poisoning. This problem made the petrochemical industry find a solution.
Detal method
So using an HF as a catalyst to provide this activation energy has one big problem. As a result, The method based on solid catalysts is
developing to replace HF acid units slowly to guarantee environmental safety and expand the economy. Many compounds such as zeolites and metal oxides have been used to make solid catalysts with selectivity, long life, economic efficiency, and high safety. Nowadays, most LAB production units use the Detal process.
The Detal process involves the dehydrogenation of linear paraffins to olefins and the side reaction with benzene using a fixed bed catalytic reactor. During the early days of LAB production, chlorination of paraffin with alkylation under AlCl3 was of particular interest. After 1960 the dehydrogenation and HF alkylation method received more attention due to its economic benefits and high quality of produced LAB. The typical paraffin dehydrogenation methods are superior to other methods due to the low cost of starting kerosene feed. About 81% of the produced alkylbenzene globally is linear based on dehydrogenation and alkylation.
So Due to the environmental problems of HF acid catalysts, the use of solid catalysts (Detal process) has thrived and has increased since 1995.
Until now, we see that in the environmental concerns and cost-effectiveness, the Detal method is way better than the HF method. But how about the quality of the produced LAB in two methods?
Comparison of the quality of linear alkene benzene produced in both methods
The quality of the two products, HF and Detal, is similar, but the LAB made from the Detal units is more linear than the linear alkylbenzene made by the HF process. But The most apparent difference between the HF and Detal methods is the higher content of 2phenylalkane in the Detal method, making the LAB better sulfonated.
New developments
Studying in the petrochemical industry to find new ways, especially using new catalysts in producing linear alkylbenzene (LAB), has continued for decades.
The cutting-edge technology is based on the Detal process called the UOP technology.
This method uses an acid-stable substrate and a noncorrosive
catalyst to substitute HF acid. But not all of them have the selectivity to produce the desired product and stability under reaction conditions.
Solid acid catalysts used for the production o fLAB must have selectivity, long life, and resilience under reaction conditions. Studies have been done on various solid catalysts such as clay, alumina, and magnesium silicate. The effect of different zeolites on the process has been investigated. The size of the zeolite has a significant impact.
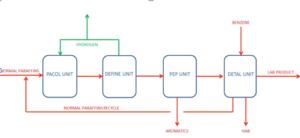
LAB plants based on UOP technology have four different units:
PACOL unit. In the unit, n-paraffins are dehydrogenated with a catalyzed reaction to produce mono-olefins.
DEFINE unit. The unit converts through a catalyst reaction di-olefins produced in the PACOL unit into mono-olefins, improving the LAB production yield and the product quality.
PEP unit. PEP process removes aromatics from the PACOL effluent stream, thus increasing the DETAL catalyst life, improving the LAB product quality, and reducing the Heavy Alkylate production.
DETAL unit. This process is a state-of-the-art process for the manufacture of detergent-grade Alkylate. It uses a solid heterogeneous catalyst to produce Linear Alkylbenzene by the direct alkylation of benzene and mono-olefins. The solid catalyst replaces the traditional liquid acid (HF acid or aluminum chloride) with significant advantages in terms of capital investment, potential environmental impact, and LAB product quality.
UOP LAB Technology has several advantages compared to standard alkylation processes.
Linear Alkyl Benzene (LAB) applications
LAB’s primary use is in the manufacture of Linear Alkyl Benzene Sulfonates (LAS). Also, LAB finds extensive application in the manufacture of domestic detergents and industrial cleaners.
Linear alkylbenzene (LAB) Effects on the
Linear alkylbenzene (LAB) Effects on the environment
Market Outlook
Just a look at the annual production process of this petrochemical product can show why the players in the petrochemical field in the tough competition are trying to gain a significant share of this market.
Let’s go back to the distant years. LAB production was about 2.8 million tons in 2002. Production of LAB in the United States is currently more than a billion pounds a year, and global usage will grow at 3.6% annually.
LAB production capacity in the world in 2018 was 4.1 million tons. An average increase of 2.2% in production capacity is forecast for 2018 to 2030. On the other hand, on average, from 2020 to 2027, growth in global demand is estimated at 4.7 percent. Last year, however, the growth was estimated at 2.7 percent, which has increased due to the prevalence of the corona.
In the global market for linear alkylbenzene, the top 5 players are Pseudo (570,000 tons), Sasol (360,000 tons), Jin Tong (350,000 tons), Fushun (280,000 tons), and Sinopec (200,000 tons).
The Middle East has about 20% of the world’s production capacity. The regional producers of this product are mainly Iranian, Saudi, and Qatari companies. Farabi Petrochemical, located in Saudi Arabia with a nominal annual capacity of 140,000 tons, is one of these companies. The company will soon inaugurate the 120,000-ton project. A company with a production capacity of 100,000 tons and a PEARL company with a production capacity of 30,000 tons are also in Qatar.
Before 1973, In Iran, the country’s need for this strategic product was met through imports. Iran Chemical Industries Investment Company was put into operation on June 1, 1994, with a capacity of 50,000 tons.
With the increase in domestic consumption and the improvement of the consumer market in the region, the implementation of the company’s development plan for the annual production of 75,000 tons of linear alkylbenzene was strengthened, and the company succeeded in completing its development plan in 2003. Also, in 2005, the establishment of Bisotun Petrochemical Company, with a production capacity of 50,000 tons, increased the country’s production capacity to 170,000 tons.
Kia Middle East commodity and energy company in Iran is one of the main players, especially in the Middle East and Central Asia. This Iranian company entered this market to respond to the improving demand for LAB but stable supply.
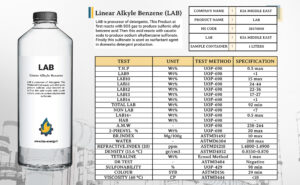
This company has full storage accessibility and a high capability of transportation. In addition to supplying Iran’s needs, it exports LAB to many countries from Asia to Europe. The company also has offices in many countries in Asia and Europe. quality of offered production is comparable with the best LAB produced. bellow we see The analysis sheet of the LAB offered by Kia Middle East Company: